Ozone Layer
Picture from WikiMedia Commons
Resources:
See NOAA for information regarding the Ozone layer!
Greenhouse Gases and Aerosols
Resources:
See NOAA for information regarding the Ozone layer!
Greenhouse Gases and Aerosols
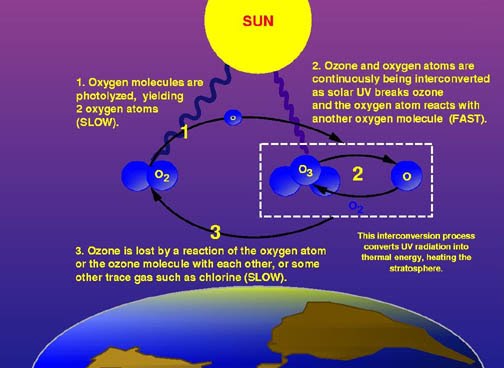
The ozone layer is a belt of naturally occuring gas it sits about 9 to 18 miles above earth and protects humans from the harmful ultraviolet B (UVB) radiation emitted by the sun. UVB exposure has been linked to skin cancer and cataracts.
The ozone layer of the earth is destroyed when chlorine and bromine atoms contact the ozone. Just one chlorine atom can destroy 100,000 ozone molecules. Some molecules that destroy the ozone layer
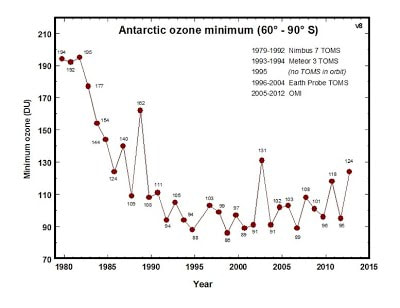
The ozone layer above the antarctic has been particularly impacted by pollution since the mid-1980s. When the sun shines for longs periods of day during the spring and summer in the south chlorine reacts with the ultraviolet rays and destroys the ozone layer up to 65 percent.
“One chlorine atom can destroy over 100,000 ozone molecules before it is removed from the stratosphere” https://www.epa.gov/ozone-layer-protection/basic-ozone-layer-science
“It sits 9.3 to 18.6 miles above Earth, and serves as a shield from the harmful ultraviolet B radiation emitted by the sun” https://www.nationalgeographic.com/environment/global-warming/ozone-depletion/
“Such deterioration allows large amounts of ultraviolet B rays to reach Earth, which can cause skin cancer and cataracts in humans and harm animals as well.”
“chlorine reacts with ultraviolet rays, destroying ozone on a massive scale, up to 65 percent.”
“scientists estimate it will take another 50 years for chlorine levels to return to their natural levels.”
“Specifically chlorine levels declined by 0.8 percent each year between 2005 and 2016.”
http://www.newsweek.com/nasa-hole-earths-ozone-layer-finally-closing-humans-did-something-771922
The ozone layer of the earth is destroyed when chlorine and bromine atoms contact the ozone. Just one chlorine atom can destroy 100,000 ozone molecules. Some molecules that destroy the ozone layer
The ozone layer above the antarctic has been particularly impacted by pollution since the mid-1980s. When the sun shines for longs periods of day during the spring and summer in the south chlorine reacts with the ultraviolet rays and destroys the ozone layer up to 65 percent.
“One chlorine atom can destroy over 100,000 ozone molecules before it is removed from the stratosphere” https://www.epa.gov/ozone-layer-protection/basic-ozone-layer-science
“It sits 9.3 to 18.6 miles above Earth, and serves as a shield from the harmful ultraviolet B radiation emitted by the sun” https://www.nationalgeographic.com/environment/global-warming/ozone-depletion/
“Such deterioration allows large amounts of ultraviolet B rays to reach Earth, which can cause skin cancer and cataracts in humans and harm animals as well.”
“chlorine reacts with ultraviolet rays, destroying ozone on a massive scale, up to 65 percent.”
“scientists estimate it will take another 50 years for chlorine levels to return to their natural levels.”
“Specifically chlorine levels declined by 0.8 percent each year between 2005 and 2016.”
http://www.newsweek.com/nasa-hole-earths-ozone-layer-finally-closing-humans-did-something-771922
Antarctic Ozone Minimum from 1980 t0 2015 (From WikiMedia Commons)